TOXIN-ANTITOXIN SYSTEMS
Toxin-antitoxin (TA) systems are a large family of genes thought to be involved in the regulation of bacterial growth and drivers of persistence. TA systems encode non-secreted toxins that inhibit essential cellular processes, and antitoxins that neutralise the toxins. It is thought that when antitoxins are degraded in response to a trigger, free toxins cause transient growth arrest through self-intoxication. We have found 14 TA systems in Salmonella, all of which to contribute positively to persistence in macrophages. This has led us to investigate how toxins of these operons function, how antitoxins neturalise their toxin partner, how the operons are regulated and what the physiological consequences of the activity of TAs are.
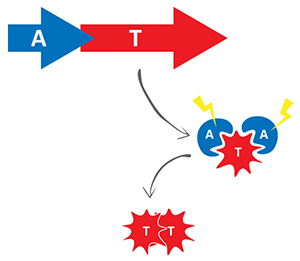
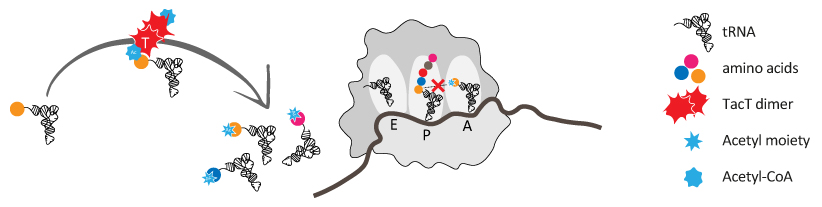
Three of the toxins are acetyltransferases that when overexpressed inhibit Salmonella growth through alteration of translation. Through structure/function analyses, we found that the TacT toxins acetylate the primary amine group of the amino acid moiety of charged
![]() RNA molecules. The addition of an acetyl moiety to the primary amine group of the aminoacyl-
RNA molecules. The addition of an acetyl moiety to the primary amine group of the aminoacyl-![]() RNA blocks its ability to support translation.
RNA blocks its ability to support translation.
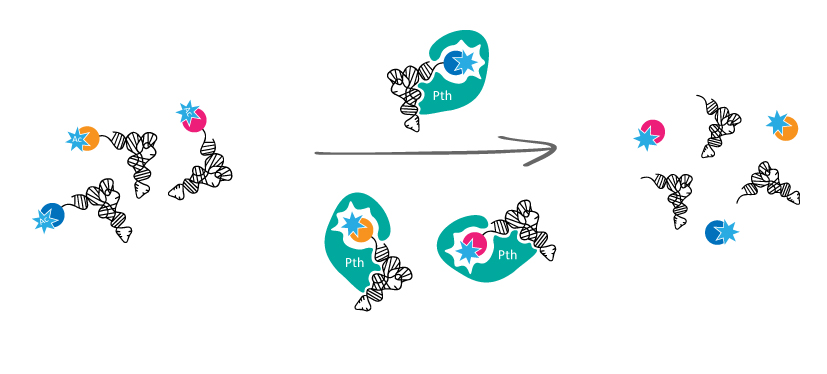
TacT-induced corruption of ![]() RNA is reversible and a detoxifying enzyme, peptidyl-
RNA is reversible and a detoxifying enzyme, peptidyl-![]() RNA hydrolase (Pth) counteracts TacT toxicity through deacylation of acetylated aa-
RNA hydrolase (Pth) counteracts TacT toxicity through deacylation of acetylated aa-![]() RNAs. The balance between Pth and TacT is critical for the level of growth-arrested persisters induced by overexpression of TacT. This suggests the existence of immunity against TA toxins beyond their cognate antitoxins.
RNAs. The balance between Pth and TacT is critical for the level of growth-arrested persisters induced by overexpression of TacT. This suggests the existence of immunity against TA toxins beyond their cognate antitoxins.