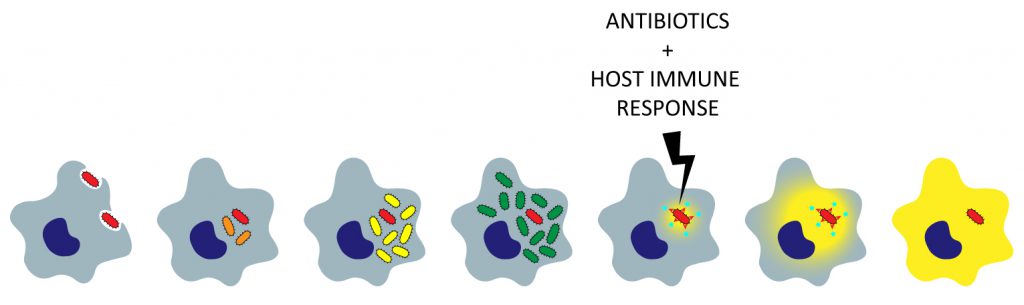
Persisters
Antibiotic persistence during infection involves the ability of persisters to survive the combined actions of drugs and host immune responses. Even though persisters have been studied for many years, little is understood about their formation and survival in a host and their effect on infection outcomes. A major focus of our research has been to develop systems to study persister biology in a host context.
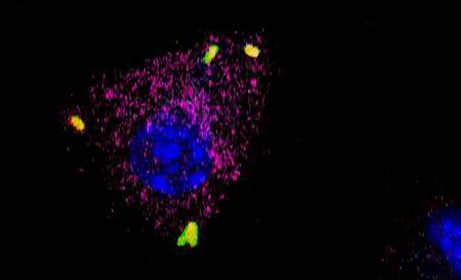
We specifically work on the intracellular pathogen Salmonella. We observed that when engulfed by host immune cells, the bacteria form high proportions of non-growing persisters. Studying the interaction between Salmonella persisters and host macrophages allows us to answer important questions about the biology of persisters.
Using fluorescent reporters of growth and dual-RNAseq, we showed that Salmonella persisters formed in macrophages display maintaine metabolic activity during infection. These Salmonella persisters, as single non-growing bacterial cells, have the remarkable capacity to reprogram the physiological state of host macrophages from a bactericidal pro-inflammatory state (M1) to a permissive anti-inflammatory (M2) phenotype. This reprogramming contributes to how persisters can survive for extended periods of time in the face of immune responses and revealed for the first time a pathogen’s ability to induce permissiveness of the host, as opposed to solely dampening the pro-inflammatory responses.